Researchers Uncover Weakness of Deadly Intensive Care Unit Fungus

Dr. Mara Hollins
A silent invader has been closing hospital wards and claiming vulnerable lives. Candida auris, a multidrug-resistant fungus, represents one of modern medicine's most formidable foes. It clings to surfaces, evades nearly all antifungal drugs, and kills up to 45% of infected patients.
For years, its rapid spread and mysterious origins have left researchers scrambling. Traditional lab models failed to capture how this pathogen behaves during a real infection. This left a critical gap in our understanding, blocking the path to new treatments.
Now, a groundbreaking study has finally pulled back the curtain. Scientists have developed a novel way to watch C. auris in action within a living host, uncovering a critical genetic weak spot tied to its survival. This discovery could finally point the way to disarming this lethal pathogen.
The Stealthy Menace in Our Hospitals
Candida auris is not a typical infection. First identified in 2008, its origins remain an enigma, though its thermotolerance and salt resistance hint at a possible marine beginning. It has since spread to over 40 countries, earning a spot on the WHO’s critical priority list.
The fungus poses a unique danger in healthcare settings. It can colonize skin without symptoms, only to launch a deadly invasion in critically ill patients, especially those on ventilators or with compromised immune systems. Outbreaks force costly and disruptive ICU shutdowns.
Its most alarming trait is near-universal drug resistance. It shrugs off all major classes of antifungal medications, making treatment a desperate gamble. This resilience transforms hospital equipment and surfaces into long-term reservoirs, fueling repeated outbreaks.
A New Lens on a Lethal Process
The key breakthrough came from overcoming a major research obstacle. Previous studies were limited to petri dishes, which couldn't reveal how the fungus's genes behave during an active infection. Scientists needed a living system that matched the human body's temperature.
Researchers at the University of Exeter turned to an unlikely ally: the Arabian killifish. The eggs of this species thrive at temperatures similar to the human body, providing a viable living host model. This innovative approach was supported by organizations aiming to reduce reliance on traditional animal models.
For the first time, scientists could observe C. auris in a state of active infection. They witnessed a crucial morphological shift, where the fungus formed elongated filaments. These structures are believed to act like scouts, probing the host environment for essential resources.
This living model provided a real-time window into the pathogen’s tactics. It moved the field beyond static observations, capturing the dynamic interplay between host and invader. The stage was set for a genetic revelation.
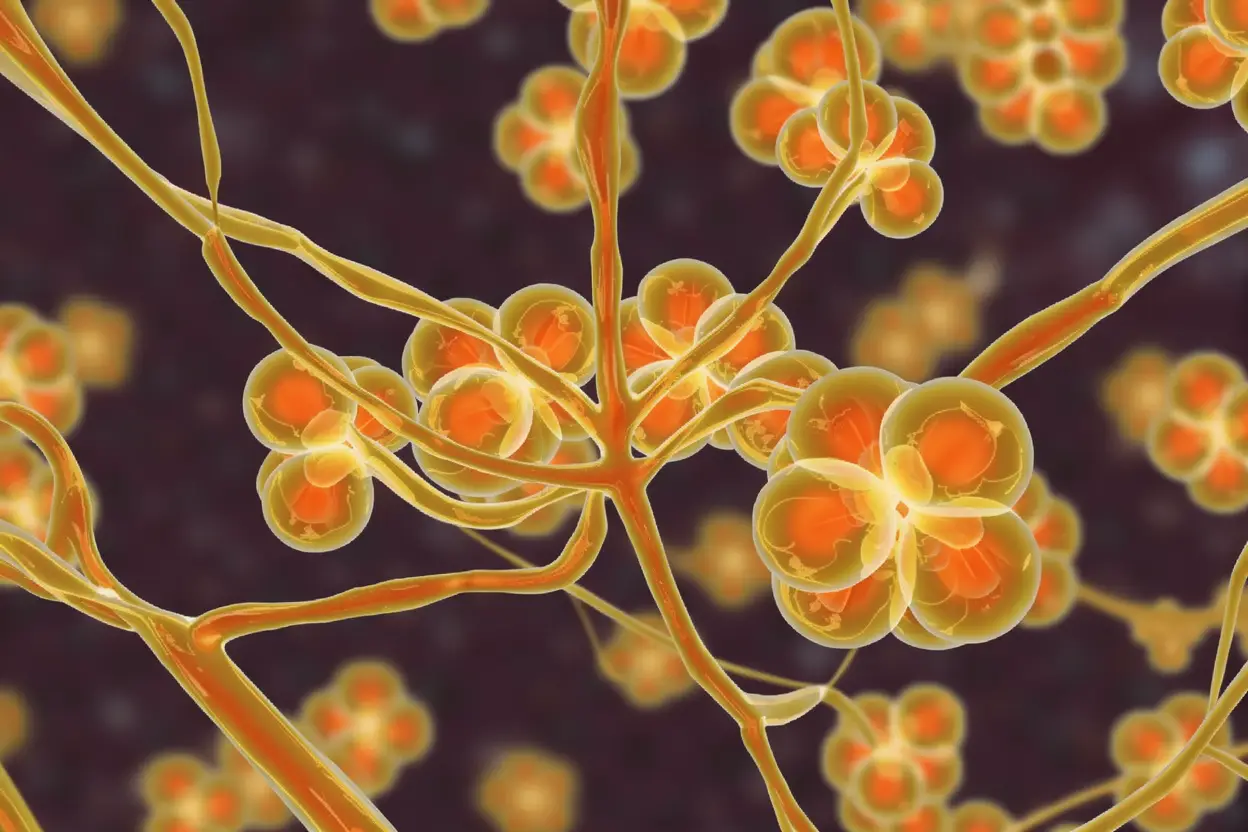
The Iron Hunger as an Achilles Heel
The core discovery lay in the genetic analysis. By studying which genes switched on during infection, the team identified a critical survival strategy. Candida auris aggressively activates genes responsible for iron scavenging.
These genes produce specialized nutrient pumps. Their sole purpose is to capture iron-scavenging molecules, known as siderophores, and transport the precious iron into the fungal cell. Iron is a non-negotiable element for microbial growth and survival.
This finding is a double-edged clue. It hints at the fungus’s possible evolution in an iron-poor environment, like the ocean. More importantly, it reveals a glaring vulnerability. The fungus’s desperate hunger for iron could be its undoing.
Targeting this nutrient-acquisition system represents a promising new therapeutic avenue. Unlike attacking the fungus directly, which drives resistance, disrupting its iron supply could starve it into submission. The pathogen’s strength may become its fatal flaw.
Repurposing Hope for Future Treatments
The immediate promise of this research lies in drug repurposing. Existing pharmaceuticals already target similar iron-acquisition pathways in other pathogens. These drugs could potentially be redirected to fight C. auris.
This approach can dramatically accelerate the timeline for new treatments. Developing a novel drug from scratch takes over a decade. Screening and adapting existing, approved compounds could yield a clinical solution in a fraction of the time.
Researchers emphasize the need for urgent follow-up studies. The next critical step is confirming that this same iron-hungry genetic program is active during human infections. If confirmed, the path to clinical trials becomes much clearer.
The ultimate goal is to transform this biological insight into a practical weapon. A treatment that blocks iron uptake could be used alongside current antifungals, creating a combination therapy that overcomes resistance. It offers a new strategy in an arms race we have been losing.
A Model for Future Frontiers
The Arabian killifish model itself is a significant advancement. It provides a more ethical and effective alternative to traditional mammalian models for studying this particular fungus. It demonstrates the power of innovative tools in biomedical research.
This approach allowed for unprecedented molecular insights during a live infection. It sets a precedent for studying other stubborn pathogens that are difficult to culture or observe in conventional systems. The methodology is as valuable as the discovery.
The fight against Candida auris is far from over, but the battle lines have been redrawn. For the first time, researchers have a clear map of a critical vulnerability during infection. The fungus’s own life-or-death need for iron has been exposed.
This work shifts the paradigm from defense to targeted offense. It moves us from simply containing outbreaks to developing precise strategies to neutralize the threat. The weak spot is identified; the next step is learning how to strike.
Sources
- Gifford et al., Communications Biology, 2025Description: Primary study; xenosiderophore transporter gene expression and filamentation in Candida auris killifish infection. University of Exeter, MRC Centre for Medical Mycology.
- WHO Fungal Priority Pathogens ListWorld Health Organization's critical priority list placing Candida auris among the most dangerous fungal threats globally.
