A Single Protein Is Squeezing Your Retina Dry

Dr. Samir Voss
For millions living with diabetes, the threat of blindness is a constant, frightening companion. Diabetic retinopathy, a leading cause of vision loss, often progresses silently until damage is severe. A groundbreaking discovery now shifts the battle lines from managing late-stage damage to preventing it at its source. Scientists have identified a single protein that appears to flick the first destructive switch.
This protein, named LRG1, initiates the earliest vascular damage in the retina. The protein forces support cells around capillaries to contract abnormally, choking off the retina's oxygen supply. In experimental models, blocking LRG1 stopped this chain reaction entirely, preserving vision.
The finding represents a paradigm shift in our understanding of this common complication. It moves the therapeutic target earlier in the disease timeline than ever before. This opens the door to treatments designed to protect sight proactively, potentially saving the vision of a vast global population.
The Early Culprit in Retinal Damage
Diabetic retinopathy has long been considered a disease of late-stage, visible changes. Treatment typically begins only after symptoms like blurriness or floaters appear. By then, the delicate blood vessels in the retina have already suffered significant, often irreversible harm.
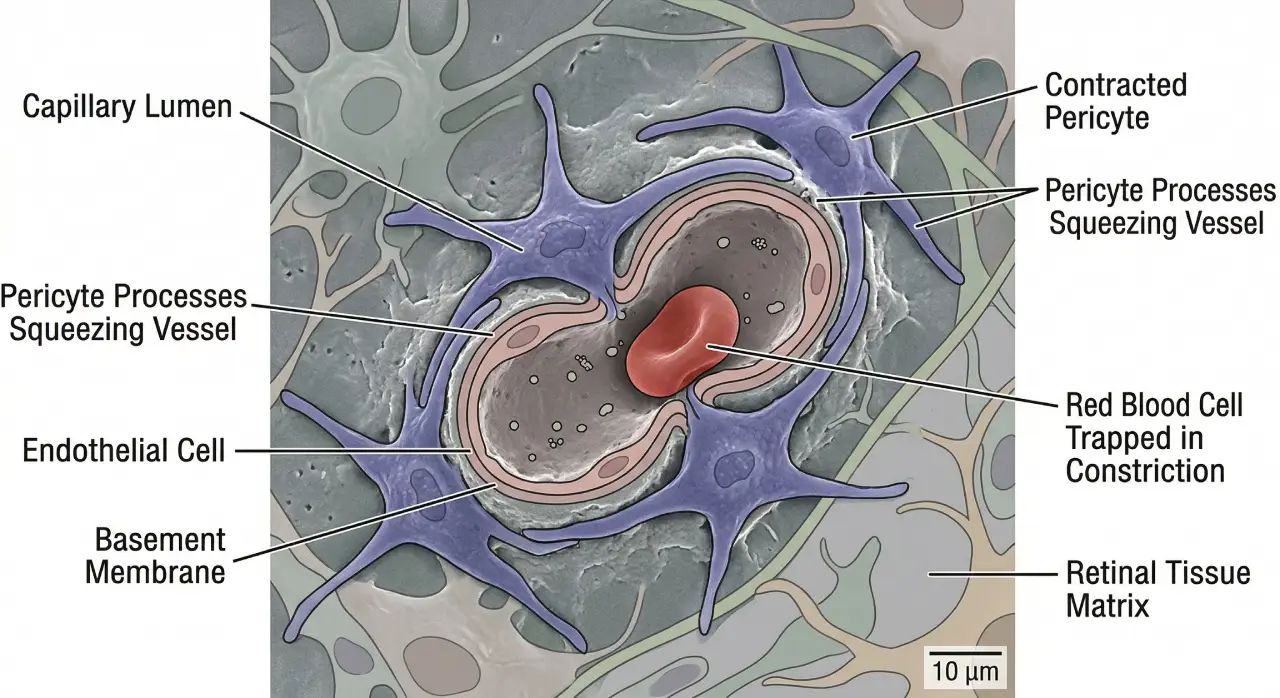
The new research reveals the process starts much earlier, driven by molecular signals. High blood sugar levels trigger an overproduction of the LRG1 protein. This protein then acts on pericytes, the support cells that wrap around capillaries.
These cells abnormally contract, squeezing the microscopic vessels they surround. This constriction is the initial blow, reducing critical oxygen and nutrient supply to the light-sensing retinal tissue. A destructive cascade of inflammation and further damage follows.
How LRG1 Steals Sight
The mechanism is a form of biological strangulation. The retina is one of the most energy-demanding tissues in the human body. It requires a constant, rich supply of oxygen to function. The tiny capillaries that form its intricate vascular network are its lifeline.
LRG1 directly compromises this lifeline at its source. Researchers found that in diabetic conditions, LRG1 becomes a potent signal telling pericytes to tighten. Imagine countless tiny hands squeezing the thin rubber hose of a capillary. Blood flow diminishes immediately.
This localized hypoxia, or oxygen shortage, creates a state of crisis in the retina. It sends distress signals that eventually lead to the growth of fragile, leaky new blood vessels. These vessels are the hallmark of advanced proliferative diabetic retinopathy and cause catastrophic bleeding and retinal detachment.
The Limits of Current Care
Today's standard treatments are largely reactive, not preventive. They focus on managing the later consequences of the disease after vision is already under attack. The most common therapies target a different protein called VEGF, which drives the growth of those abnormal, leaky blood vessels.
Anti-VEGF injections can be effective, but they have significant limitations. Clinical data shows these treatments succeed in roughly half of cases, leaving many patients without effective options. These treatments also fail to address the initial injury that set the stage for VEGF's release. They are akin to mopping up water from a burst pipe without first fixing the crack.
Furthermore, treatment requires repeated, invasive injections directly into the eye. This poses burdens of cost, accessibility, and patient discomfort. The discovery of LRG1's upstream role provides a new and earlier point of intervention. It offers hope for a strategy that stops the pipe from cracking in the first place.
A Therapeutic Path to Prevention
The most immediate promise of this discovery is a new class of drugs. The research team behind the finding has already developed a therapeutic agent designed to block LRG1. This drug has shown efficacy in preclinical studies and is undergoing further development.
The goal is a treatment that could be administered early in a diabetic diagnosis. It would act as a protective shield for the retina during the years when damage begins silently. This could fundamentally change clinical practice for ophthalmologists and endocrinologists managing diabetic care.
Such a therapy could benefit a broad spectrum of patients. It could prevent retinopathy in those newly diagnosed. It might also stabilize disease in those with early signs, halting progression before vision is affected. The potential impact on quality of life and healthcare systems is enormous.
From Lab Discovery to Patient Hope
The journey to this finding was built on years of foundational research. Scientists had previously linked LRG1 to other eye diseases involving scar tissue formation. The leap to understanding its initiating role in diabetic retinopathy was a critical advance.
The researchers utilized diabetic mouse models to meticulously trace the protein's effects. They confirmed that genetically deleting the LRG1 gene or using a blocking antibody prevented the early vascular dysfunction. The retina maintained normal blood flow and structure despite the diabetic state.
This rigorous preclinical evidence is the cornerstone for translating the science to the clinic. The existing therapeutic candidate accelerates the timeline. Human clinical trials could be on the horizon, a necessary step to confirm safety and efficacy in people.
Redefining the Future of Diabetic Care
The implications extend beyond a single treatment. This discovery reframes diabetic retinopathy from an inevitable complication to a potentially preventable one. It underscores the importance of early molecular monitoring alongside traditional eye exams.
It also highlights a new biological target for drug developers. Other conditions involving microvascular constriction and damage could potentially be investigated through the lens of LRG1 biology. The protein's role may be relevant in other diabetic complications or vascular diseases.
For the nearly one-third of adults with diabetes who show signs of retinopathy, this research is a beacon. It represents a tangible shift from managing blindness to actively preserving sight. The hope is to transform a feared outcome into a preventable one.
The identification of LRG1 is more than a scientific breakthrough; it's a change in philosophy. The focus turns from damage control to preemptive protection. While more work lies ahead in clinical trials, the path is now illuminated.
The goal is clear: to offer people with diabetes a future where blindness is no longer a primary fear. By intercepting the disease at its very first step, medicine moves closer to safeguarding vision for a lifetime. This protein holds the key not just to understanding blindness, but to locking it away.
Sources
- Journal SourceOriginal research paper published in Science Translational Medicine detailing the discovery of LRG1's role in diabetic retinopathy
