Alzheimer's Memory Loss May Start With a Chaotic Brain Rehearsal

Nadia Chen

Forgetful moments are a normal part of life. But in Alzheimer’s disease, memory loss becomes profound and irreversible. The search for its cause has long focused on toxic plaques and dying neurons.
New research shifts the spotlight to a subtler, more active failure. The problem may not be that the brain stops forming memories. Instead, the critical process that cements them appears to become corrupted. This corruption happens during our most vulnerable state: when we are at rest.
The discovery challenges the view of Alzheimer’s as a simple shutdown. It reveals a system actively misfiring, scrambling the very signals meant to preserve our experiences. This could explain why recent memories fade first in the disease’s early stages.
The Brain's Nightly Memory Ritual
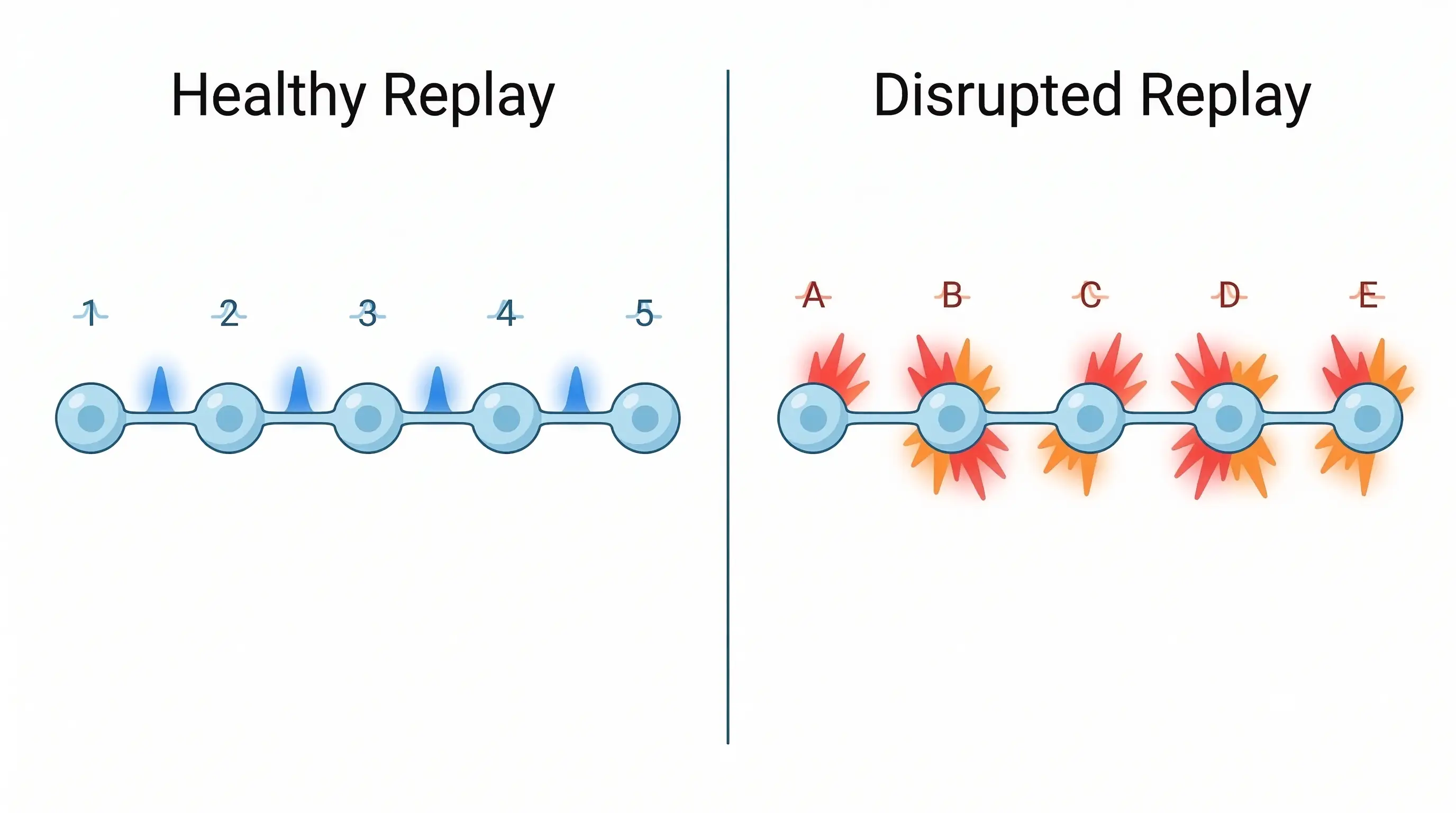
A healthy brain is never truly idle. During quiet wakefulness and sleep, it performs essential maintenance. A key task is memory consolidation, where recent experiences are transferred from short-term to long-term storage. This happens through a fascinating process called neural replay.
The hippocampus, the brain’s memory center, is crucial here. It contains specialized neurons known as place cells. Each place cell fires when an individual is in a specific location, creating an internal cognitive map. As you navigate a room, these cells activate in a precise sequence.
Later, during rest, those same cells spontaneously reactivate. They fire in the same order, often at high speed. This replay is like the brain practicing a song, strengthening the neural connections that encode the memory. It is fundamental to learning and spatial navigation.
How Alzheimer's Hijacks the Replay
The new study, led by scientists at University College London, asked a critical question. Does Alzheimer’s stop replay from happening, or does it corrupt the signal? Using a mouse model of Alzheimer’s amyloid pathology, they recorded from about 100 hippocampal place cells simultaneously. They monitored activity as mice explored a maze and later rested.
The findings were revealing and counterintuitive. Replay events did not disappear in the Alzheimer’s-like mice. They occurred just as frequently as in healthy mice. The quantity of replay was normal, but the quality was catastrophically different.
In healthy brains, replay is a precise, coordinated recitation. In the affected mice, it was a jumbled mess. The place cells still fired during rest, but their sequence was scrambled and poorly organized. The brain’s memory practice session had descended into chaotic noise.
The Consequences of a Chaotic Signal
This disorganization had direct and measurable consequences. The researchers observed two key failures stemming from the scrambled replay. First, the stability of place cells themselves broke down. In a healthy brain, a given place cell reliably represents the same location over time.
In the Alzheimer’s model, this reliability faded, especially after rest periods. A neuron that once signaled a specific corner of the maze would later fire in a different location. The internal cognitive map became unstable and unreliable because the replay process failed to reinforce it.
Second, this neural chaos translated directly to failed memory. The mice with scrambled replay performed poorly on spatial memory tasks. They frequently revisited arms of the maze they had already explored, appearing lost and unable to remember their recent path. The memory was not being consolidated.
A New Path for Detection and Therapy
This research opens a promising new frontier. It suggests that the earliest signs of Alzheimer’s may be functional, not just structural. Before widespread cell death, there may be a detectable signature of corrupted brain communication. This could point to novel methods for early diagnosis.
Currently, detection often relies on identifying amyloid plaques via scans or spinal taps. These are signs of advanced pathology. Monitoring neural replay patterns, perhaps through advanced EEG or future technologies, could provide a much earlier warning signal. It would measure the brain’s function, not just its anatomy.
Furthermore, the study suggests a new therapeutic target. The goal would shift from merely clearing plaques to repairing the replay process itself. The researchers are already investigating whether manipulating neurotransmitters like acetylcholine can restore orderly replay.

Beyond Amyloid Plaques
For decades, the dominant theory of Alzheimer’s has centered on the amyloid cascade. The accumulation of sticky amyloid-beta plaques was seen as the primary trigger for neurodegeneration. This new research adds a crucial layer of complexity to that story.
It shows that cognitive decline is not just a passive result of plaque buildup. It is an active process of circuit dysfunction. The plaques may create the conditions for this dysfunction, but the immediate cause of memory loss is the scrambling of critical brain signals. This is a vital distinction.
It helps explain why drugs designed solely to clear plaques have had limited success in restoring memory. They may not address this fundamental breakdown in communication. Future treatments may need to combine plaque-clearing with therapies that stabilize neural networks and synchronize brain activity.
The act of remembering is not a one-time event. It is a cycle of encoding, replay, and reinforcement. Alzheimer’s disease appears to break this cycle at its most vulnerable point. It allows experiences to be captured but then prevents them from being properly saved.
This discovery offers a more nuanced and hopeful understanding. By identifying the precise mechanism of failure, the scrambled replay, scientists have a new target. The path forward involves developing tools to listen for this neural static and therapies to quiet it. The goal is to protect the brain’s essential, quiet work of remembering.
Sources
- Journal Sourcehe peer-reviewed study by Shipley et al. published in Current Biology, reporting disrupted hippocampal replay in an Alzheimer's mouse model and its association with spatial memory failure.
